Experimental Technique
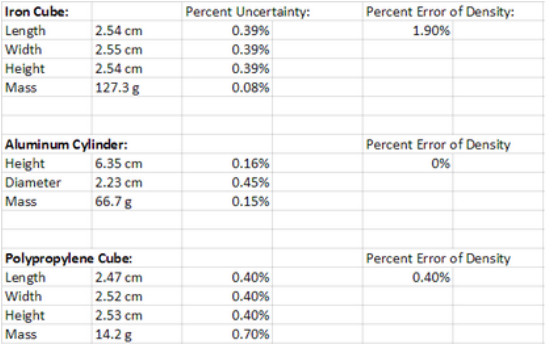
Data
Analysis
Example Equations
Volume:
Volume=Length x Width x Height
Volume = 2.54cm x 2.55cm x 2.54cm
Volume = 16.5cm^3
Density:
Density = Mass / Volume
Density = 127.3g / 16.5cm^3
Density = 7.72g/cm^3
Percent Uncertainty:
Percent Uncertainty = Uncertainty / Magnitude x 100%
Percent Uncertainty = 0.1cm^3 / 16.5 cm^3 x 100%
Percent Uncertainty = 0.62%
Percent Error:
Percent Error = (|Accepted Value - Measured Value| / Accepted Value) x 100%
Percent Error = (|7.72-7.87| / 7.87) x 100%
Percent Error = 1.9%
Conclusion
My results for the density of the iron cube, aluminum cylinder, and polypropylene cube were very close to the actual results. The iron cube I measured the iron cubes density at 7.72g/cm^3 while the actual density was 7.87g/cm^3. This was a 1.9 percent error. The aluminum cylinder I measured to have a density of 2.70g/cm^3 and the actual density of aluminum was 2.7g/cm^3. I had a 0 percent error in this measurement. The polypropylene cubes actually density was 0.9g/cm^3 and i had measured 0.904g/cm^3. This was a 0.4 percent error. Some of my errors could have been caused by the rounded edges of the cubes and the slight variations on the cube sizes. A way to get a better measurement would be to have a perfect cube with sharp edges.
My results for the density of the iron cube, aluminum cylinder, and polypropylene cube were very close to the actual results. The iron cube I measured the iron cubes density at 7.72g/cm^3 while the actual density was 7.87g/cm^3. This was a 1.9 percent error. The aluminum cylinder I measured to have a density of 2.70g/cm^3 and the actual density of aluminum was 2.7g/cm^3. I had a 0 percent error in this measurement. The polypropylene cubes actually density was 0.9g/cm^3 and i had measured 0.904g/cm^3. This was a 0.4 percent error. Some of my errors could have been caused by the rounded edges of the cubes and the slight variations on the cube sizes. A way to get a better measurement would be to have a perfect cube with sharp edges.